In the last few years, the landscape of weight loss has shifted seismic plates. For decades, the prevailing advice was a variation of “eat less, move more”—a model that framed obesity almost exclusively as a failure of willpower. But the meteoric rise of Semaglutide (sold under brand names like Ozempic, Wegovy, and Rybelsus) has fundamentally changed that conversation.
It is no longer just about calories in versus calories out; it is about hormones, signaling pathways, and the biological architecture of hunger.
Semaglutide is not a stimulant, a fat burner, or a diuretic. It is a peptide that hacks the body’s own satiety system. But how exactly does a weekly injection cause people to lose 15% to 20% of their body weight? The answer lies in a complex interplay between the gut, the brain, and the pancreas.
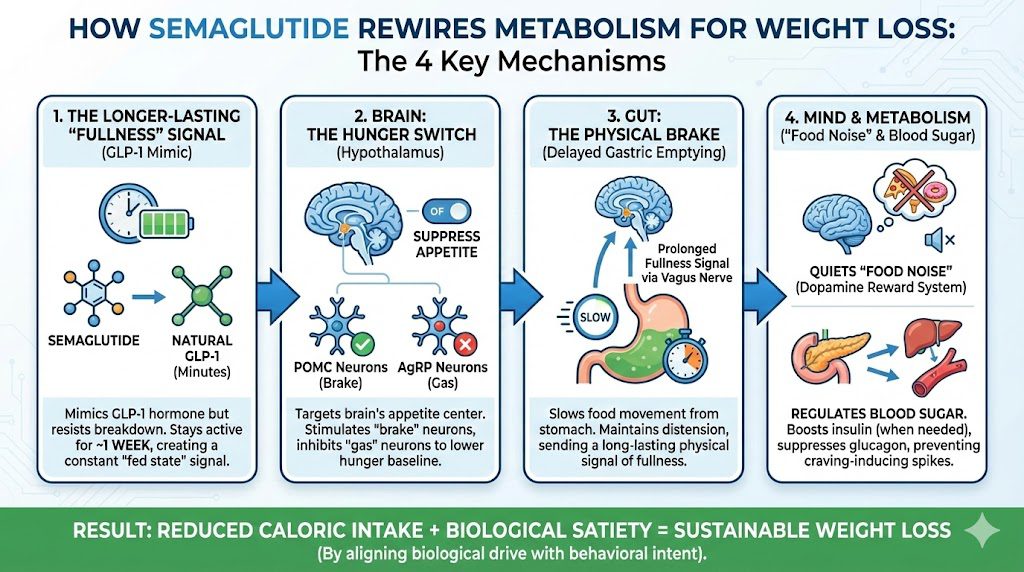
1. The Biological Mimic: Understanding GLP-1
To understand Semaglutide, you have to understand the hormone it copies: Glucagon-Like Peptide-1 (GLP-1).
Under normal conditions, when you eat a meal, your intestines release a cascade of hormones. One of the most important is GLP-1. This hormone travels through the bloodstream and acts as a biological messenger, delivering a “fed state” signal to various organs. It tells the pancreas to release insulin, tells the liver to stop dumping sugar into the blood, and tells the brain that you have had enough to eat.
The Problem with Nature: Natural GLP-1 is incredibly short-lived. It has a half-life of only 1 to 2 minutes before it is broken down by an enzyme called DPP-4. It provides a brief signal of fullness, and then it vanishes.
The Semaglutide Solution: Semaglutide is a synthetic agonist (mimic) of GLP-1. However, it has been chemically modified to survive the DPP-4 enzyme. Instead of lasting two minutes, Semaglutide has a half-life of approximately one week. This means that once injected, it keeps the body in a state of heightened “fullness signaling” 24 hours a day, 7 days a week, rather than just for a few minutes after a meal.
2. Mechanism I: The Hypothalamus (The Hunger Switch)
The most profound effect of Semaglutide occurs in the brain. The drug crosses the blood-brain barrier and binds to GLP-1 receptors in the hypothalamus, the control center for appetite and metabolism.
Within the hypothalamus, there are two primary sets of neurons that regulate weight:
- POMC Neurons: These are the “brake pedal.” When activated, they suppress appetite and increase energy expenditure.
- AgRP Neurons: These are the “gas pedal.” When activated, they trigger intense hunger and food-seeking behavior.
Semaglutide essentially keeps its foot on the brake. By stimulating the POMC neurons and inhibiting the AgRP neurons, it drastically lowers the body’s “hunger baseline.” Users often report forgetting to eat or feeling satisfied after consuming portions that are half the size of what they previously ate. It physically limits the desire for food before the first bite is even taken.
3. Mechanism II: Delayed Gastric Emptying
While the brain effect reduces the desire to eat, the gastric effect reduces the ability to overeat.
GLP-1 receptors are highly concentrated in the stomach. When Semaglutide binds to these receptors, it slows down gastric motility—the muscular contractions that push food from the stomach into the small intestine.
Normally, the stomach might empty a meal in 1 to 2 hours. On Semaglutide, this process is significantly decelerated. Because food remains in the stomach for a much longer duration, physical distension is maintained. This sends a prolonged mechanical signal of fullness to the brain via the vagus nerve.
This mechanism is why overeating on Semaglutide can be physically painful. If a user attempts to eat their pre-drug portion sizes, the food has nowhere to go, leading to nausea, bloating, and vomiting.
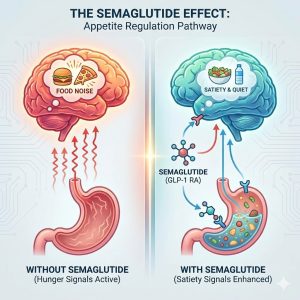
4. Mechanism III: The “Food Noise” Silencer
Perhaps the most fascinating—and least understood—aspect of Semaglutide is its psychological impact. Many people living with obesity describe a phenomenon known as Food Noise.
What is Food Noise? It is the constant intrusive mental chatter about food: What am I eating for lunch? Is there leftover cake in the breakroom? I shouldn’t have eaten that. When is dinner?
Semaglutide appears to affect the brain’s dopaminergic reward system (the mesolimbic pathway). This is the same pathway involved in addiction. By dampening the dopamine spike associated with palatable foods, Semaglutide turns the volume down on food noise.
Users report that food becomes neutral. A slice of pizza looks like a slice of pizza, not a source of comfort or excitement. This liberation from obsessive food thoughts allows people to make dietary choices based on logic rather than compulsion.
5. Mechanism IV: Metabolic Regulation (Insulin & Glucagon)
While weight loss is the headline, Semaglutide was originally designed for Type 2 Diabetes, and its metabolic effects are critical for fat loss.
- Insulin Secretion: It stimulates the pancreas to release insulin only when blood sugar is high. Insulin helps usher glucose into cells to be used for energy.
- Glucagon Suppression: It inhibits the release of glucagon, a hormone that tells the liver to release stored sugar into the bloodstream.
By stabilizing blood sugar levels, Semaglutide prevents the spikes and crashes that often drive sugar cravings. When blood sugar remains stable, energy levels stay consistent and the body is less likely to trigger emergency hunger signals.
6. The Clinical Reality: Results vs. Risks
Semaglutide is not magic; it is a tool that amplifies the effects of a caloric deficit.
The Efficacy Data: In the landmark STEP trials, participants taking 2.4 mg weekly lost an average of 14.9% of their body weight over 68 weeks, compared to just 2.4% in the placebo group.
The Side Effect Profile:
- GI Distress: Because it slows the stomach, nausea, vomiting, diarrhea, and constipation are common.
- Muscle Loss (Sarcopenia): Rapid weight loss often includes muscle loss without adequate protein and resistance training.
- “Ozempic Face”: Rapid facial fat loss can create a gaunt or aged appearance.
7. The “Set Point” Theory and Long-Term Use
The Set Point Theory suggests the body fights to maintain a certain weight range. When you diet, metabolism slows and hunger hormones increase.
Semaglutide appears to lower this set point, convincing the body it is safe to release stored energy without triggering starvation alarms.
The Rebound Trap: However, current data suggests Semaglutide is a treatment rather than a cure. When the drug is discontinued, gastric emptying speeds up and food noise often returns. Many users regain up to two-thirds of lost weight within a year.
Conclusion
Semaglutide helps people lose weight not by burning fat directly but by altering the signals the brain receives about hunger and energy balance. It creates a physiological environment where eating less feels natural rather than forced. By synchronizing the gut and brain, it bridges the gap between biological drive and behavioral intent.

Leave a comment
Your email address will not be published. Required fields are marked *
You must be logged in to post a comment.